QUESTION IMAGE
Question
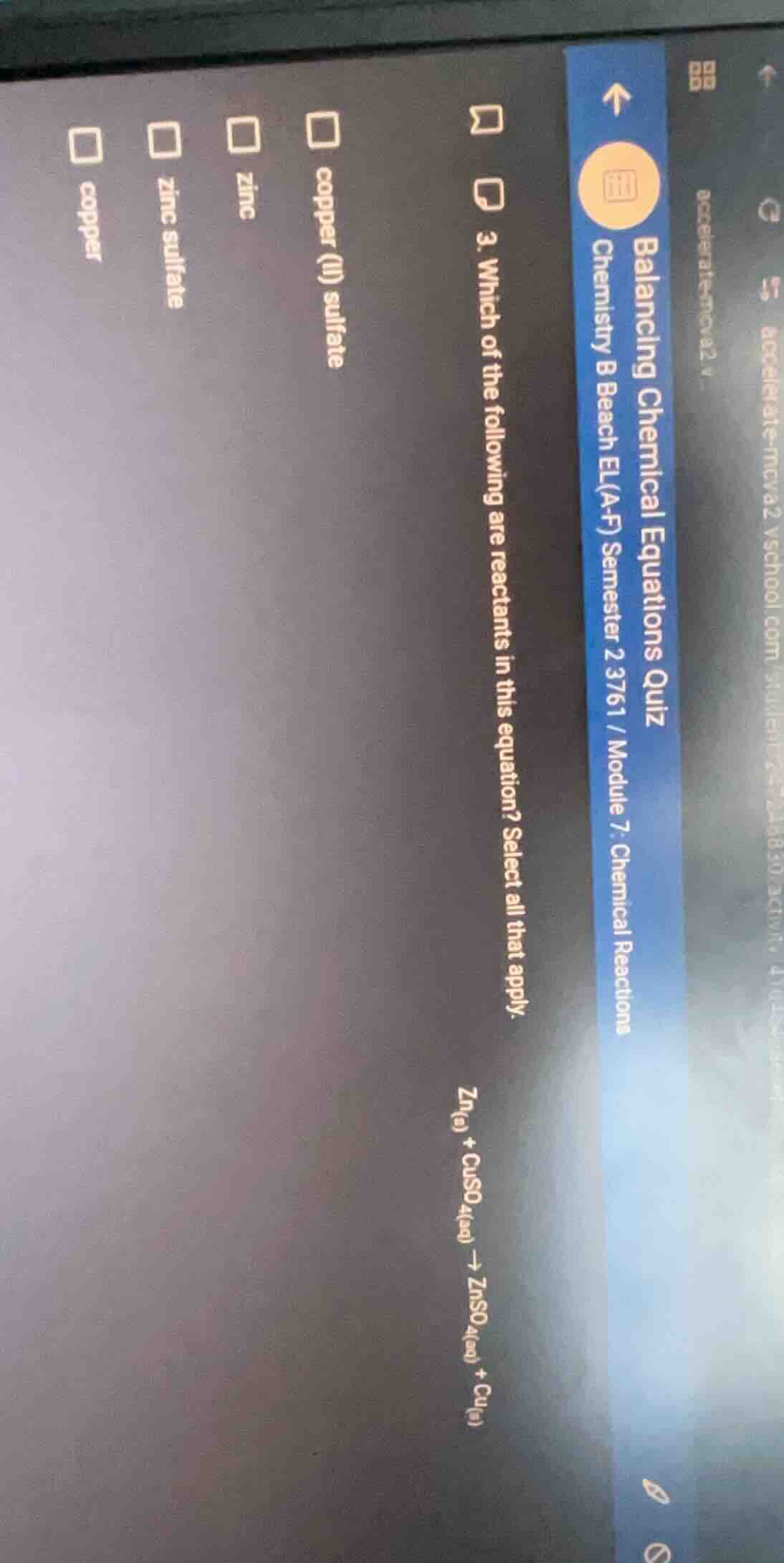
- which of the following are reactants in this equation? select all that apply.
zn₍ₛ₎ + cuso₄₍ₐq₎ → znso₄₍ₐq₎ + cu₍ₛ₎
options: copper (ii) sulfate, zinc, zinc sulfate, copper
In a chemical equation, reactants are the substances that undergo a reaction and are located on the left - hand side of the arrow. The given chemical equation is $\ce{Zn_{(s)} + CuSO_{4(aq)} -> ZnSO_{4(aq)} + Cu_{(s)}}$. So we need to identify which of the given substances are on the left - hand side (reactants). $\ce{Zn}$ (zinc) and $\ce{CuSO_{4}}$ (copper (II) sulfate) are the reactants. We check the options: "zinc" corresponds to $\ce{Zn}$, "copper (II) sulfate" corresponds to $\ce{CuSO_{4}}$. The other options like "zinc sulfate" and "copper" are products (on the right - hand side of the arrow), and "copper" (the other one) is also a product.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- zinc
- copper (II) sulfate