QUESTION IMAGE
Question
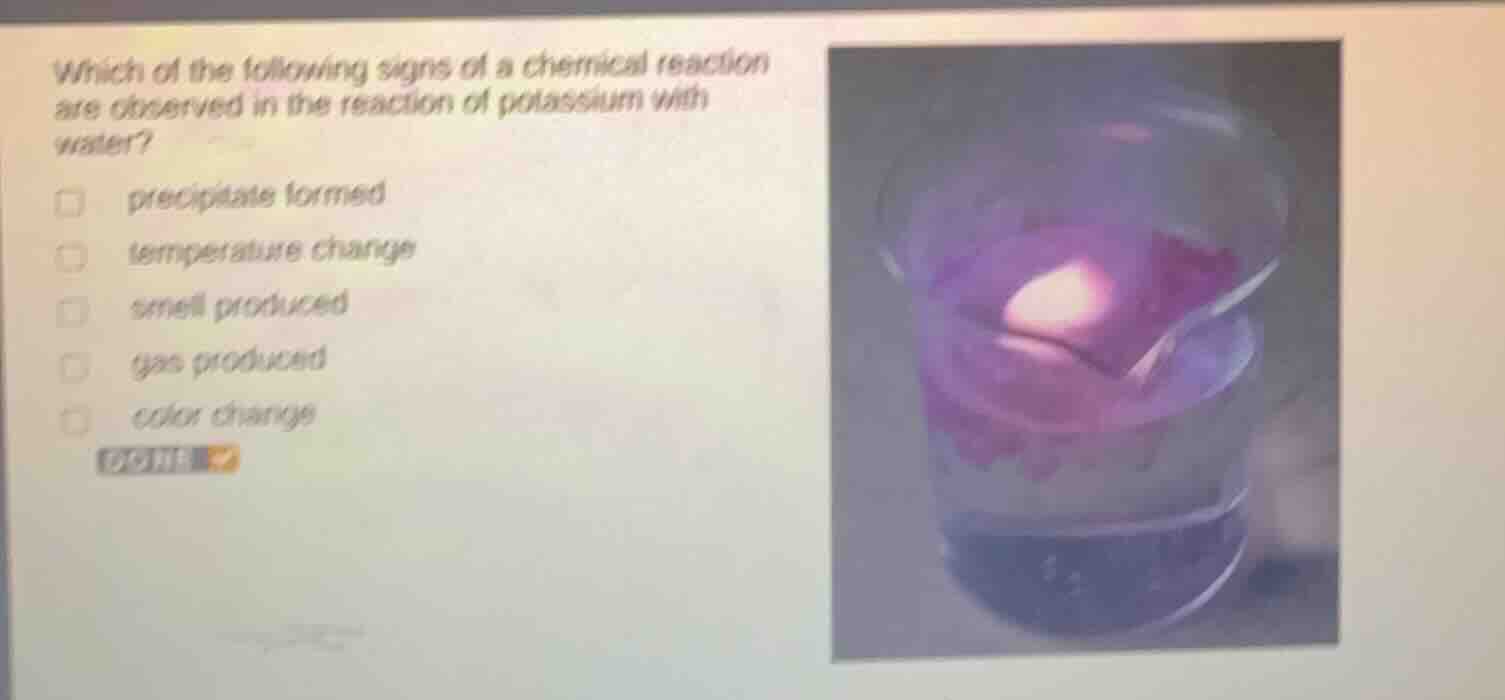
which of the following signs of a chemical reaction are observed in the reaction of potassium with water? precipitate formed temperature change smell produced gas produced color change
Brief Explanations
- Precipitate formed: Potassium reacts with water to form \(KOH\) (potassium hydroxide) which is soluble, so no precipitate.
- Temperature change: The reaction \(2K + 2H_2O=2KOH + H_2\uparrow\) is highly exothermic, so temperature increases (temperature change observed).
- Smell produced: \(KOH\) is odorless in this reaction context, and \(H_2\) is odorless, so no distinct smell.
- Gas produced: The reaction produces \(H_2\) (hydrogen gas), so gas is produced.
- Color change: The solution turns pink/purple (due to phenolphthalein - like indication from the basic \(KOH\) and maybe the reaction's visual aspects with potassium), so color change is observed.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- temperature change (the reaction is exothermic, so temperature increases)
- gas produced (hydrogen gas is released: \(2K + 2H_2O = 2KOH+H_2\uparrow\))
- color change (the solution becomes pink/purple due to the formation of basic \(KOH\) and the reaction's visual effects with potassium)