QUESTION IMAGE
Question
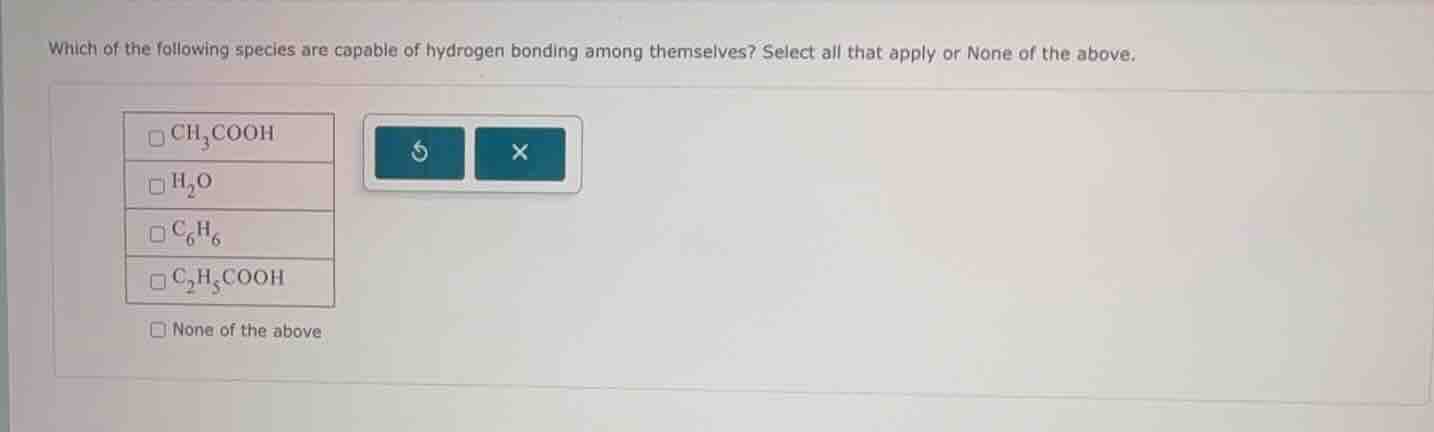
which of the following species are capable of hydrogen bonding among themselves? select all that apply or none of the above. \\(\square\ce{ch3cooh}\\) \\(\square\ce{h2o}\\) \\(\square\ce{c6h6}\\) \\(\square\ce{c2h5cooh}\\) \\(\square\\)none of the above
Brief Explanations
To determine if a species can form hydrogen bonds, it must have a hydrogen atom bonded to a highly electronegative atom (F, O, or N) and a lone pair on F, O, or N.
- $\ce{CH_{3}COOH}$ (acetic acid) has -OH group (H bonded to O), so can form H - bonds.
- $\ce{H_{2}O}$ has H bonded to O, so can form H - bonds.
- $\ce{C_{6}H_{6}}$ (benzene) has H bonded to C (not electronegative enough), so no H - bonds.
- $\ce{C_{2}H_{5}COOH}$ (propanoic acid) has -OH group (H bonded to O), so can form H - bonds.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\ce{CH_{3}COOH}$, $\ce{H_{2}O}$, $\ce{C_{2}H_{5}COOH}$