QUESTION IMAGE
Question
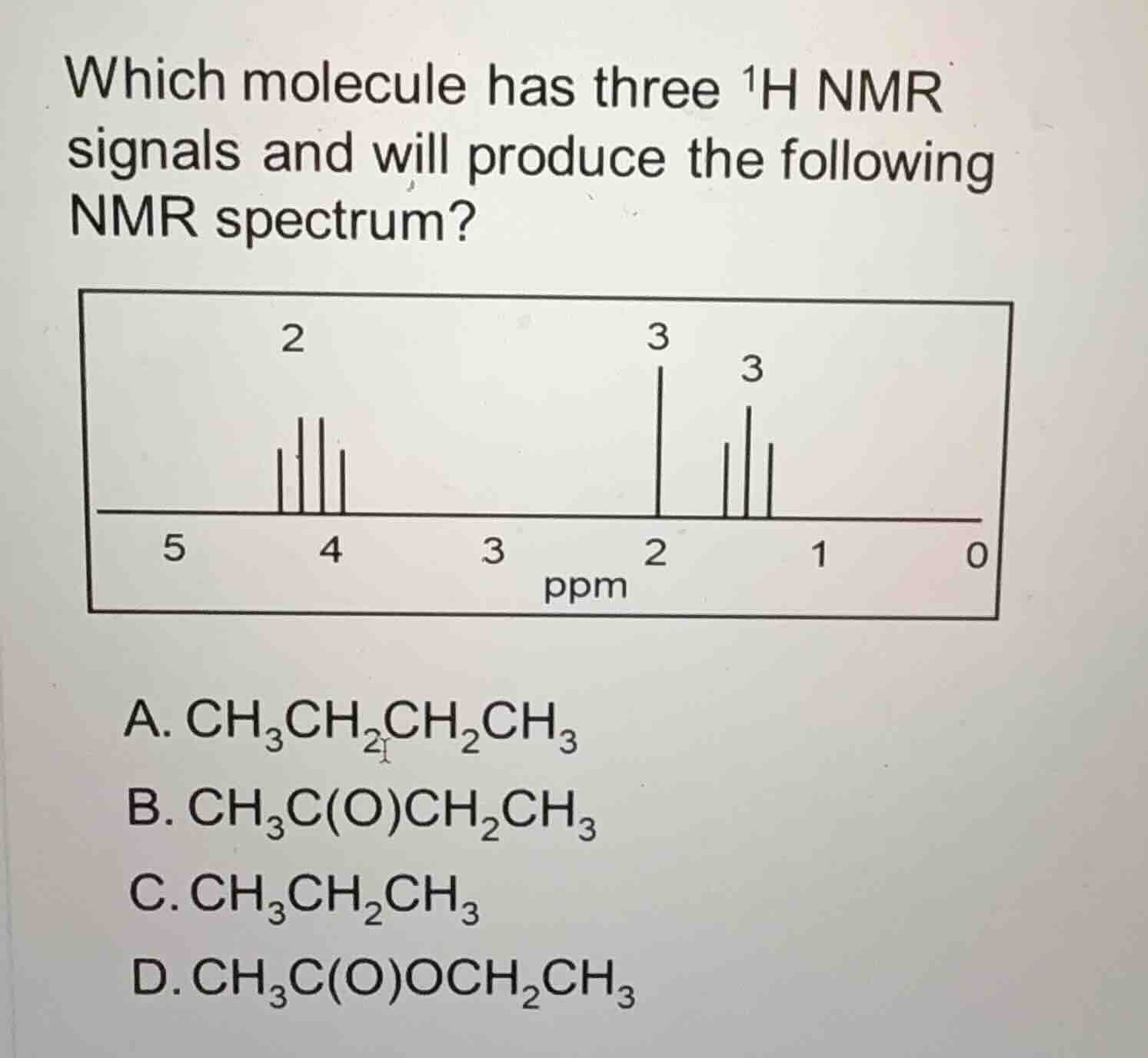
which molecule has three $^1$h nmr signals and will produce the following nmr spectrum?
a. $\ce{ch_3ch_2ch_2ch_3}$
b. $\ce{ch_3c(o)ch_2ch_3}$
c. $\ce{ch_3ch_2ch_3}$
d. $\ce{ch_3c(o)och_2ch_3}$
Step1: Analyze each option's proton environments
- Option A: \( \text{CH}_3\text{CH}_2\text{CH}_2\text{CH}_3 \) (butane) has two types of protons: \( \text{CH}_3 \) (end groups) and \( \text{CH}_2 \) (middle groups). So it should have 2 \( ^1\text{H} \) NMR signals. Eliminate A.
- Option B: \( \text{CH}_3\text{C(O)CH}_2\text{CH}_3 \) (butanone). The protons: \( \text{CH}_3 \) (adjacent to C=O), \( \text{CH}_2 \) (adjacent to \( \text{CH}_3 \) and C=O), and \( \text{CH}_3 \) (end group). Wait, let's check environments: \( \text{CH}_3 \) (C=O adjacent: 3 H), \( \text{CH}_2 \) (between \( \text{CH}_3 \) and C=O: 2 H), \( \text{CH}_3 \) (end: 3 H). Wait, but let's check the integration. The NMR spectrum has integrations 2, 3, 3. Wait, no, let's re - check Option D.
- Option C: \( \text{CH}_3\text{CH}_2\text{CH}_3 \) (propane) has two types of protons: \( \text{CH}_3 \) (end) and \( \text{CH}_2 \) (middle). So 2 \( ^1\text{H} \) NMR signals. Eliminate C.
- Option D: \( \text{CH}_3\text{C(O)OCH}_2\text{CH}_3 \) (ethyl acetate). The proton environments:
- \( \text{CH}_3 \) (adjacent to C=O: 3 H, integration 3)
- \( \text{CH}_2 \) (adjacent to O: 2 H, integration 2)
- \( \text{CH}_3 \) (end of ethyl group: 3 H, integration 3)
Now check the chemical shifts: The \( \text{CH}_2 \) adjacent to O is deshielded (around 4 ppm, which matches the multiplet with integration 2), the \( \text{CH}_3 \) adjacent to C=O is around 2 ppm (integration 3), and the end \( \text{CH}_3 \) is around 1 - 2 ppm (integration 3, and a triplet? Wait, the ethyl group's \( \text{CH}_3 \) is a triplet (adjacent to \( \text{CH}_2 \)), and the \( \text{CH}_2 \) is a quartet (adjacent to \( \text{CH}_3 \)). The \( \text{CH}_3 \) adjacent to C=O is a singlet? Wait, no, in ethyl acetate, the \( \text{CH}_3 \) (C=O - \( \text{CH}_3 \)) is a singlet (no adjacent protons), the \( \text{CH}_2 \) (O - \( \text{CH}_2 \)) is a quartet (adjacent to \( \text{CH}_3 \)), and the \( \text{CH}_3 \) (ethyl - \( \text{CH}_3 \)) is a triplet. The integrations are 3 (C=O - \( \text{CH}_3 \)), 2 (O - \( \text{CH}_2 \)), 3 (ethyl - \( \text{CH}_3 \)), which matches the spectrum's integrations (2, 3, 3? Wait, the spectrum has integrations 2 (for \( \text{CH}_2 \)), 3 (for \( \text{CH}_3 \) adjacent to C=O), 3 (for ethyl - \( \text{CH}_3 \))? Wait, maybe I mixed up the integration order. The \( \text{CH}_2 \) (O - \( \text{CH}_2 \)) has integration 2, the \( \text{CH}_3 \) (C=O - \( \text{CH}_3 \)) has integration 3, and the ethyl - \( \text{CH}_3 \) has integration 3. The chemical shift of \( \text{O - CH}_2 \) is around 4 ppm (deshielded by O), which matches the multiplet at ~4 ppm with integration 2. The \( \text{C=O - CH}_3 \) is around 2 ppm (integration 3), and the ethyl - \( \text{CH}_3 \) is around 1 - 2 ppm (integration 3, triplet).
Option B: In \( \text{CH}_3\text{C(O)CH}_2\text{CH}_3 \), the \( \text{CH}_2 \) is adjacent to C=O and \( \text{CH}_3 \). The \( \text{CH}_2 \) protons would be at a different shift. The \( \text{CH}_3 \) adjacent to C=O is at ~2 ppm, \( \text{CH}_2 \) at ~2 - 3 ppm, and \( \text{CH}_3 \) at ~1 ppm. But the integration for \( \text{CH}_2 \) in B is 2, \( \text{CH}_3 \) (C=O) is 3, \( \text{CH}_3 \) (end) is 3. Wait, but the chemical shift of \( \text{CH}_2 \) in B is not around 4 ppm. In ethyl acetate (Option D), the \( \text{CH}_2 \) is adjacent to O, so it's deshielded (higher ppm, around 4 ppm), which matches the multiplet at ~4 ppm. So Option D has three proton environments with integrations 2 (O - \( \text{CH}_2 \)), 3 (C=O - \( \te…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. \( \text{CH}_3\text{C(O)OCH}_2\text{CH}_3 \)