QUESTION IMAGE
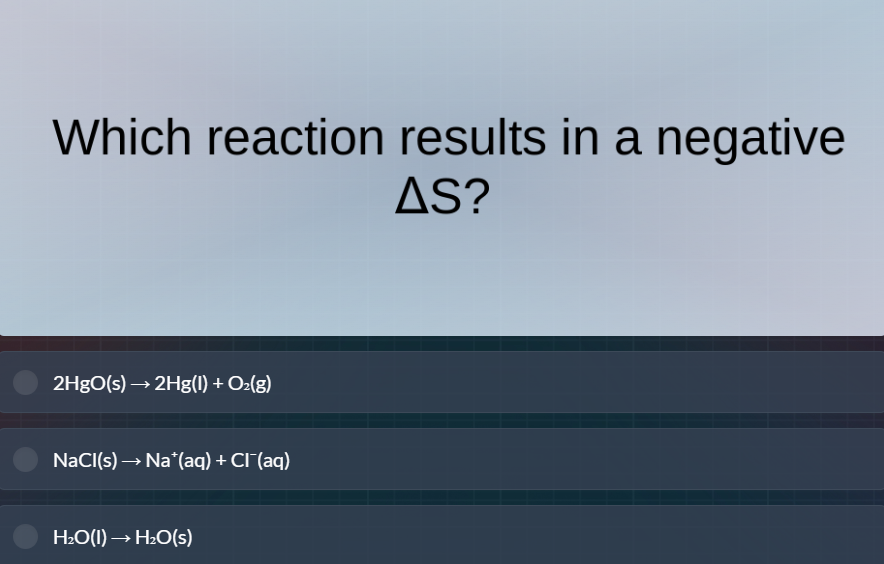
Question
which reaction results in a negative δs? 2hgo(s)→2hg(l) + o₂(g) nacl(s)→na⁺(aq) + cl⁻(aq) h₂o(l)→h₂o(s)
To determine which reaction has a negative \(\Delta S\) (entropy change), we analyze the change in disorder (entropy) for each reaction:
Step 1: Recall Entropy Trends
Entropy (\(S\)) generally increases with:
- Phase changes: \( \text{solid}
ightarrow \text{liquid}
ightarrow \text{gas} \) (more disorder).
- Formation of more moles of gas (or particles) from fewer.
- Dissolution (solids dissolving into ions in solution, increasing disorder).
Step 2: Analyze Each Reaction
- **\( 2\text{HgO}(s)
ightarrow 2\text{Hg}(l) + \text{O}_2(g) \)**
- Reactant: solid (\( \text{HgO} \)).
- Products: liquid (\( \text{Hg} \)) and gas (\( \text{O}_2 \)).
- Phase change: solid → liquid + gas (more disorder).
- Moles of gas: 0 → 1 (increase).
- \(\Delta S > 0\) (entropy increases).
- **\( \text{NaCl}(s)
ightarrow \text{Na}^+(aq) + \text{Cl}^-(aq) \)**
- Reactant: solid (\( \text{NaCl} \)).
- Products: aqueous ions (\( \text{Na}^+ \), \( \text{Cl}^- \)).
- Dissolution: solid → ions in solution (more disorder).
- \(\Delta S > 0\) (entropy increases).
- **\( \text{H}_2\text{O}(l)
ightarrow \text{H}_2\text{O}(s) \)**
- Reactant: liquid (\( \text{H}_2\text{O}(l) \)).
- Product: solid (\( \text{H}_2\text{O}(s) \)).
- Phase change: liquid → solid (less disorder, more ordered).
- \(\Delta S < 0\) (entropy decreases).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(\boldsymbol{\text{H}_2\text{O}(l)
ightarrow \text{H}_2\text{O}(s)}\) (the third option)