QUESTION IMAGE
Question
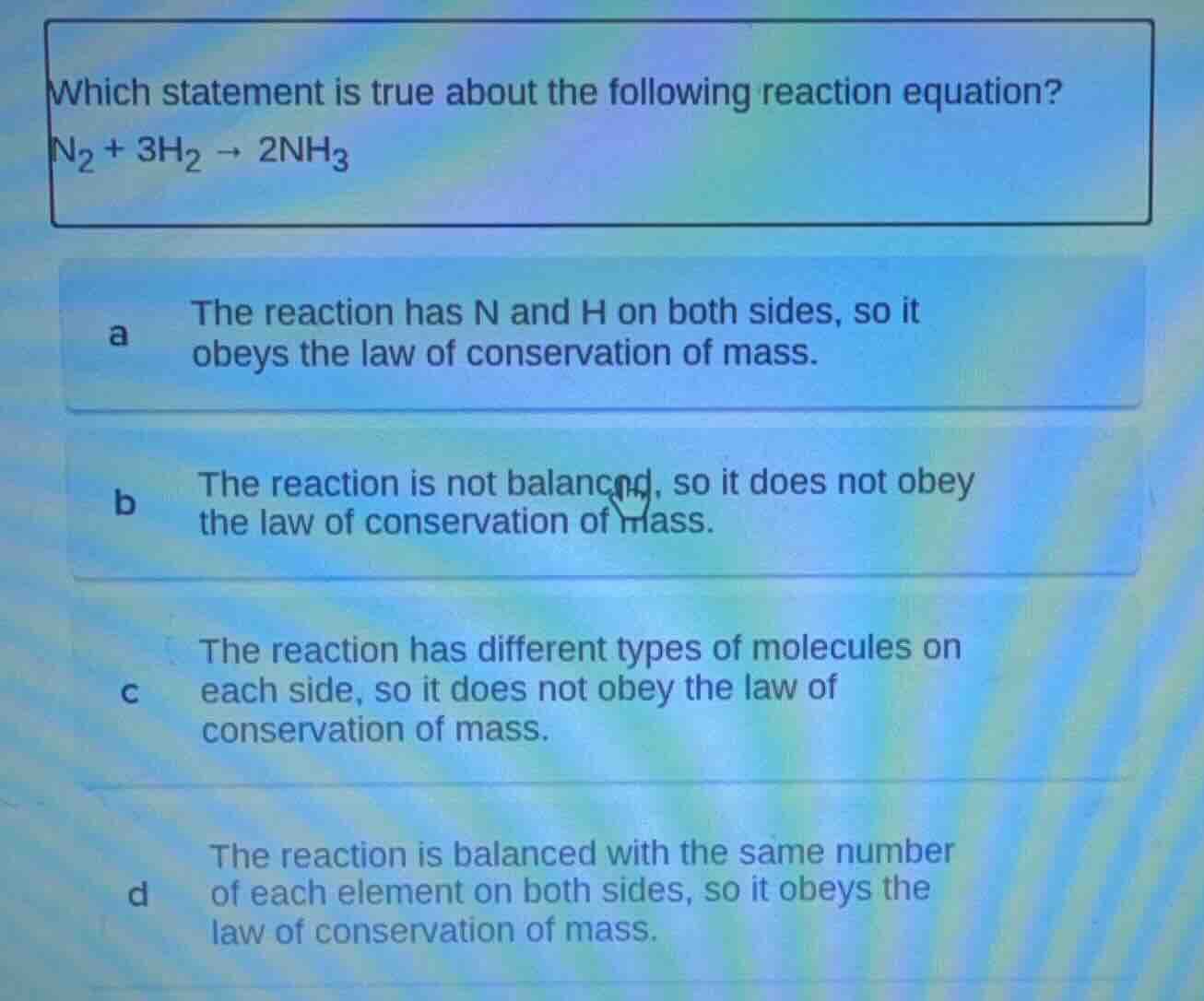
which statement is true about the following reaction equation?
$\ce{n_{2} + 3h_{2} -> 2nh_{3}}$
a the reaction has n and h on both sides, so it
obeys the law of conservation of mass.
b the reaction is not balanced, so it does not obey
the law of conservation of mass.
c the reaction has different types of molecules on
each side, so it does not obey the law of
conservation of mass.
d the reaction is balanced with the same number
of each element on both sides, so it obeys the
law of conservation of mass.
To determine the correct statement, we analyze the reaction \( \ce{N2 + 3H2 -> 2NH3} \) and the law of conservation of mass (mass is conserved in a chemical reaction, meaning the number of each atom is the same on both sides).
- Option a: Just having N and H on both sides isn’t enough—we need the same number of each atom. The law of conservation of mass requires balanced atom counts, not just presence. Eliminate a.
- Option b: The reaction is balanced. On the left: 2 N (from \( \ce{N2} \)) and 6 H (from \( 3\ce{H2} \)). On the right: 2 N (from \( 2\ce{NH3} \)) and \( 2 \times 3 = 6 \) H. So it is balanced. Eliminate b.
- Option c: Different molecules (reactants vs. products) are normal in reactions. The law of conservation of mass depends on atom counts, not molecule types. Eliminate c.
- Option d: The reaction is balanced (2 N and 6 H on both sides), so it obeys the law of conservation of mass. This matches the requirements of the law.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
d. The reaction is balanced with the same number of each element on both sides, so it obeys the law of conservation of mass.