QUESTION IMAGE
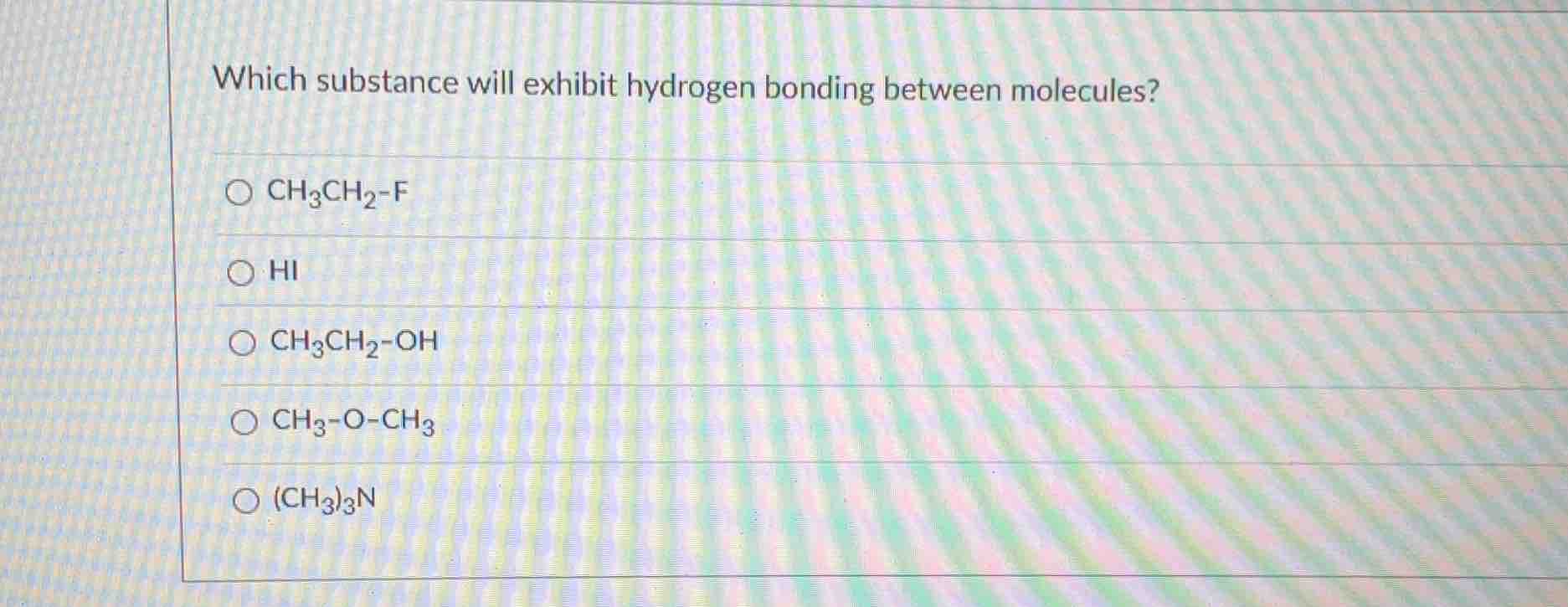
Question
which substance will exhibit hydrogen bonding between molecules?
○ $\ce{ch3ch2-f}$
○ $\ce{hi}$
○ $\ce{ch3ch2-oh}$
○ $\ce{ch3-o-ch3}$
○ $\ce{(ch3)3n}$
To determine which substance exhibits hydrogen bonding, we use the rule: Hydrogen bonding occurs when a molecule has a hydrogen atom bonded to a highly electronegative atom (F, O, or N) and can interact with another electronegative atom (F, O, or N) in a neighboring molecule.
- \( \text{CH}_3\text{CH}_2\text{-F} \): The F is bonded to C, not H, so no H - F bond for H - bonding.
- \( \text{HI} \): I is not highly electronegative (F, O, N are required), so no H - bonding.
- \( \text{CH}_3\text{CH}_2\text{-OH} \): Has an -OH group (H bonded to O). The H in -OH can form H - bonds with O atoms in neighboring \( \text{CH}_3\text{CH}_2\text{-OH} \) molecules.
- \( \text{CH}_3\text{-O-CH}_3 \): O is bonded to C, no H bonded to O, so no H - bonding.
- \( (\text{CH}_3)_3\text{N} \): N is bonded to C, no H bonded to N, so no H - bonding.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( \boldsymbol{\text{CH}_3\text{CH}_2\text{-OH}} \) (the option with the structure \( \text{CH}_3\text{CH}_2\text{-OH} \))