QUESTION IMAGE
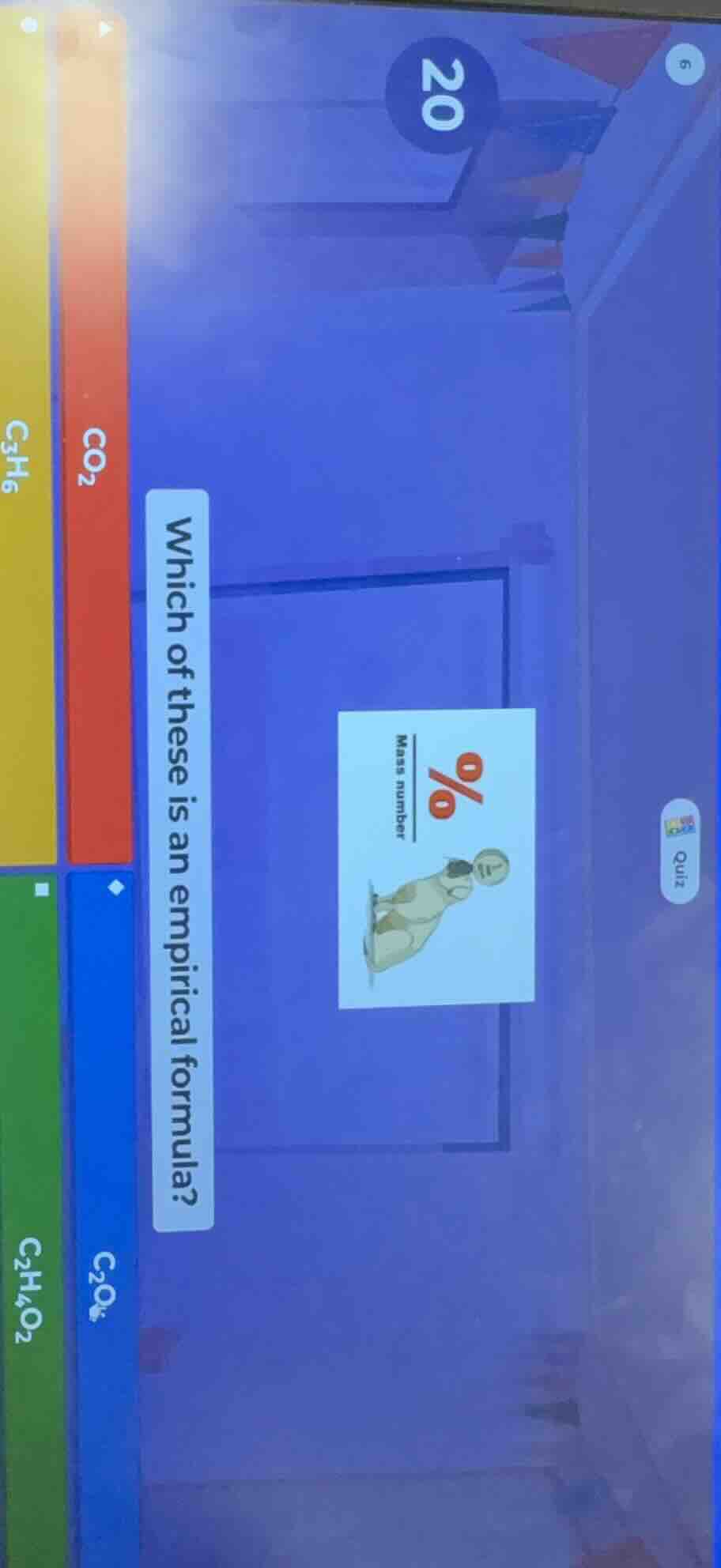
Question
which of these is an empirical formula?
$ce{co_{2}}$
$ce{c_{3}h_{6}}$
$ce{c_{2}o_{4}}$
$ce{c_{2}h_{4}o_{2}}$
Step1: Define empirical formula
An empirical formula is the simplest whole-number ratio of atoms in a compound.
Step2: Analyze $\text{CO}_2$
The ratio C:O is 1:2, which cannot be simplified further with whole numbers.
Step3: Analyze $\text{C}_3\text{H}_6$
The ratio C:H is 3:6, which simplifies to 1:2, so this is a molecular formula, not empirical.
Step4: Analyze $\text{C}_2\text{O}_4$
The ratio C:O is 2:4, which simplifies to 1:2, so this is a molecular formula, not empirical.
Step5: Analyze $\text{C}_2\text{H}_4\text{O}_2$
The ratio C:H:O is 2:4:2, which simplifies to 1:2:1, so this is a molecular formula, not empirical.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\text{CO}_2$