QUESTION IMAGE
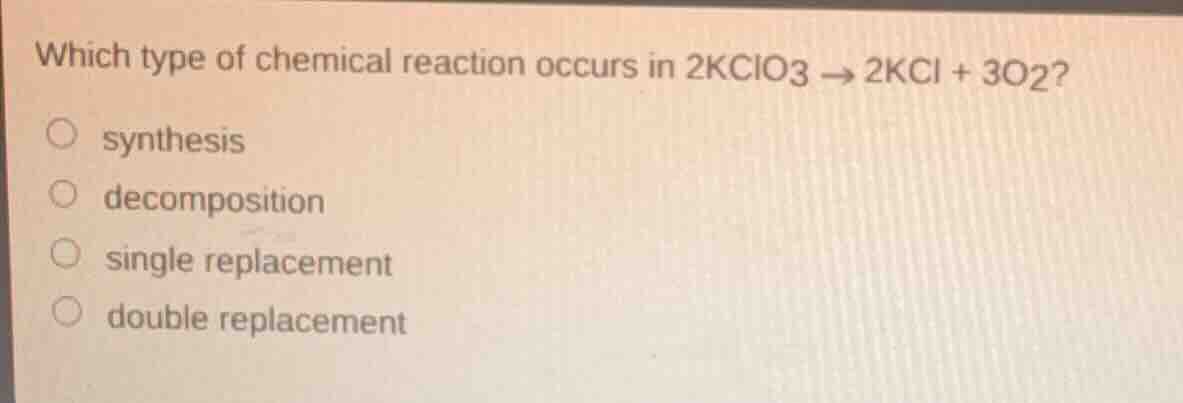
Question
which type of chemical reaction occurs in 2kclo3 → 2kcl + 3o2?
○ synthesis
○ decomposition
○ single replacement
○ double replacement
To determine the reaction type, we analyze the equation \( 2KClO_3
ightarrow 2KCl + 3O_2 \). A decomposition reaction is when a single compound breaks down into two or more simpler substances. Here, potassium chlorate (\( KClO_3 \)) breaks down into potassium chloride (\( KCl \)) and oxygen (\( O_2 \)), fitting the decomposition definition. Synthesis reactions combine substances, single replacement involves one element replacing another in a compound, and double replacement involves two compounds exchanging ions—none of these match here.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. decomposition