QUESTION IMAGE
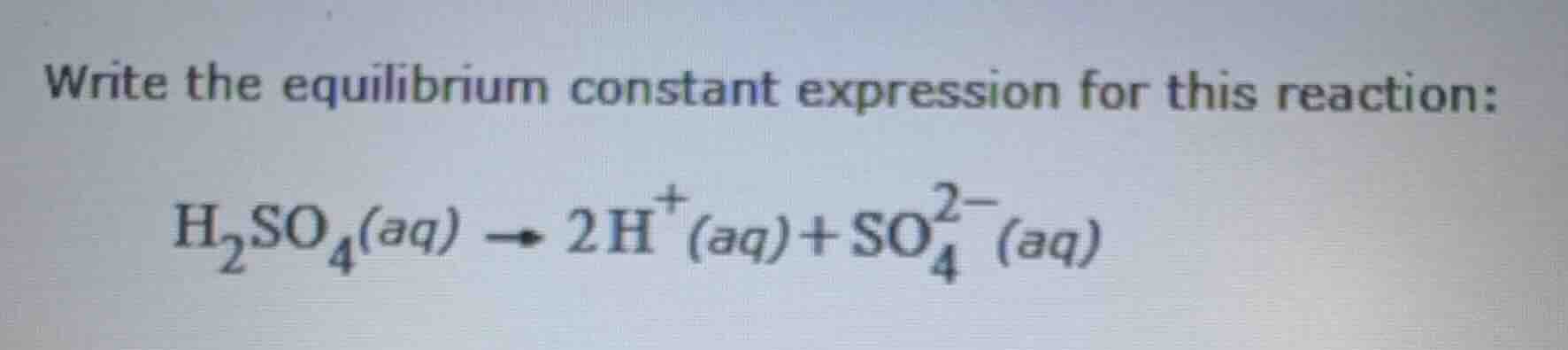
Question
write the equilibrium constant expression for this reaction: h₂so₄(aq) → 2h⁺(aq) + so₄²⁻(aq)
Step1: Recall the equilibrium constant formula
The equilibrium constant expression \( K_{eq} \) (or \( K_c \)) for a reaction \( aA + bB
ightleftharpoons cC + dD \) is \( K_{eq}=\frac{[C]^c[D]^d}{[A]^a[B]^b} \), where the concentrations of pure solids and liquids are considered as 1 (not included). For aqueous solutions, we use their molar concentrations. In the given reaction \( \ce{H2SO4(aq)
ightleftharpoons 2H+(aq) + SO4^{2-}(aq)} \) (assuming it's an equilibrium, though \( \ce{H2SO4} \) is a strong acid, but for the sake of writing the expression), the reactant is \( \ce{H2SO4(aq)} \) and products are \( \ce{H+(aq)} \) and \( \ce{SO4^{2-}(aq)} \).
Step2: Apply the formula to the reaction
For the reaction \( \ce{H2SO4(aq)
ightleftharpoons 2H+(aq) + SO4^{2-}(aq)} \), the equilibrium constant expression will be the product of the concentrations of the products raised to their stoichiometric coefficients divided by the concentration of the reactant raised to its stoichiometric coefficient. The stoichiometric coefficient of \( \ce{H2SO4} \) is 1, for \( \ce{H+} \) it is 2, and for \( \ce{SO4^{2-}} \) it is 1. So, \( K = \frac{[\ce{H+}]^2[\ce{SO4^{2-}}]}{[\ce{H2SO4}]} \) (Note: For strong acids like \( \ce{H2SO4} \), the dissociation is nearly complete, but the equilibrium constant expression is written in the same way as per the formula for the reaction as given).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( K=\frac{[\ce{H+}]^2[\ce{SO4^{2-}}]}{[\ce{H2SO4}]} \)