QUESTION IMAGE
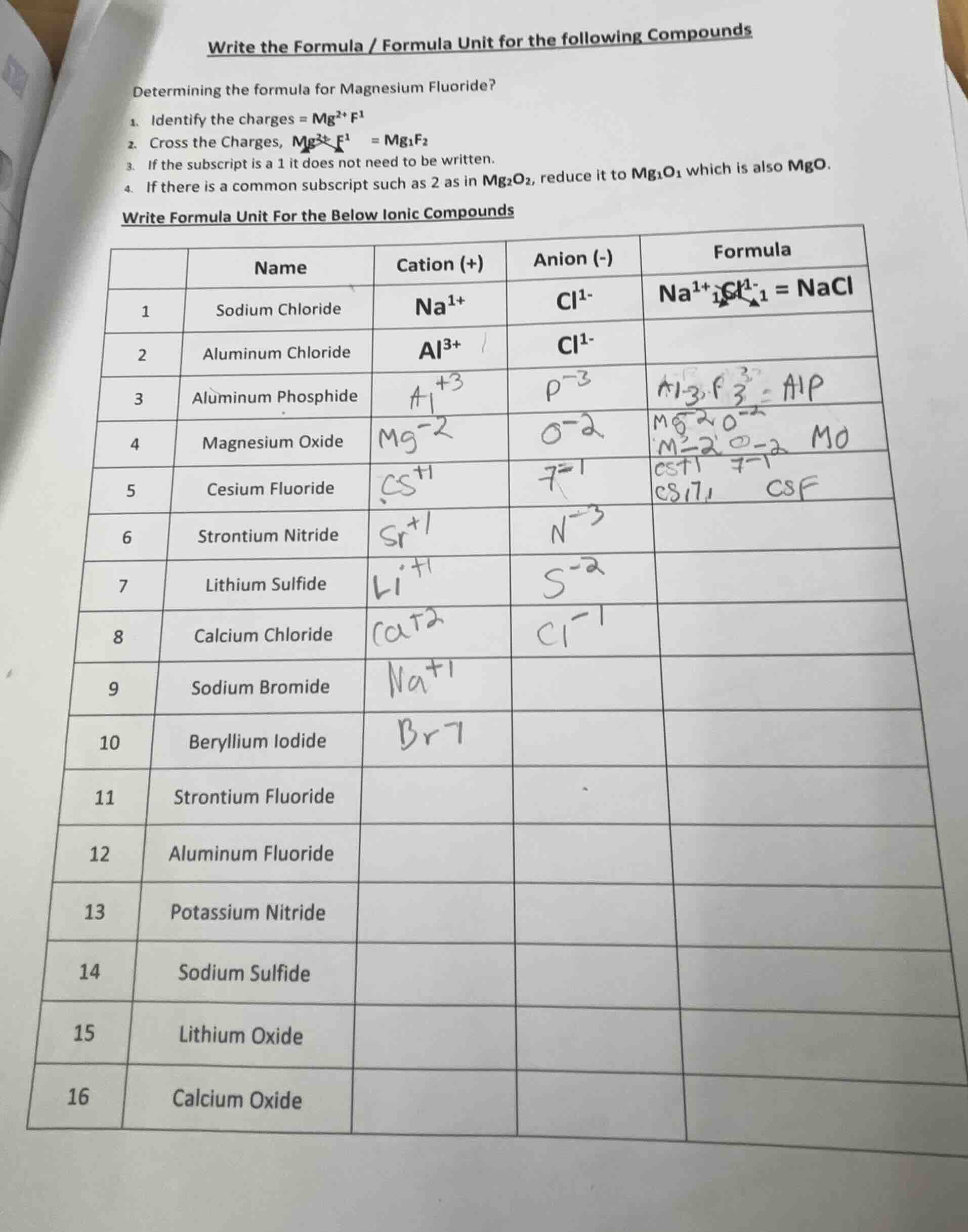
Question
write the formula / formula unit for the following compounds
determining the formula for magnesium fluoride?
- identify the charges = mg²⁺ f¹
- cross the charges, mg²⁺ f¹ = mg₁f₂
- if the subscript is a 1 it does not need to be written.
- if there is a common subscript such as 2 as in mg₂o₂, reduce it to mg₁o₁ which is also mgo.
write formula unit for the below ionic compounds
| name | cation (+) | anion (-) | formula | |
| 1 | sodium chloride | na¹⁺ | cl¹⁻ | na¹⁺cl¹⁻ = nacl |
| 2 | aluminum chloride | al³⁺ | cl¹⁻ | |
| 3 | aluminum phosphide | |||
| 4 | magnesium oxide | |||
| 5 | cesium fluoride | |||
| 6 | strontium nitride | |||
| 7 | lithium sulfide | |||
| 8 | calcium chloride | |||
| 9 | sodium bromide | |||
| 10 | beryllium iodide | |||
| 11 | strontium fluoride | |||
| 12 | aluminum fluoride | |||
| 13 | potassium nitride | |||
| 14 | sodium sulfide | |||
| 15 | lithium oxide | |||
| 16 | calcium oxide |
Step1: Cross charges for AlCl₃
$\text{Al}^{3+} \text{Cl}^{1-}
ightarrow \text{Al}_1\text{Cl}_3 = \text{AlCl}_3$
Step2: Correct Sr charge, cross for Sr₃N₂
$\text{Sr}^{2+} \text{N}^{3-}
ightarrow \text{Sr}_3\text{N}_2$
Step3: Cross charges for Li₂S
$\text{Li}^{1+} \text{S}^{2-}
ightarrow \text{Li}_2\text{S}_1 = \text{Li}_2\text{S}$
Step4: Cross charges for CaCl₂
$\text{Ca}^{2+} \text{Cl}^{1-}
ightarrow \text{Ca}_1\text{Cl}_2 = \text{CaCl}_2$
Step5: Identify anion, cross for NaBr
$\text{Na}^{1+} \text{Br}^{1-}
ightarrow \text{NaBr}$
Step6: Correct cation, cross for BeI₂
$\text{Be}^{2+} \text{I}^{1-}
ightarrow \text{Be}_1\text{I}_2 = \text{BeI}_2$
Step7: Identify ions, cross for SrF₂
$\text{Sr}^{2+} \text{F}^{1-}
ightarrow \text{Sr}_1\text{F}_2 = \text{SrF}_2$
Step8: Identify ions, cross for AlF₃
$\text{Al}^{3+} \text{F}^{1-}
ightarrow \text{Al}_1\text{F}_3 = \text{AlF}_3$
Step9: Identify ions, cross for K₃N
$\text{K}^{1+} \text{N}^{3-}
ightarrow \text{K}_3\text{N}_1 = \text{K}_3\text{N}$
Step10: Identify ions, cross for Na₂S
$\text{Na}^{1+} \text{S}^{2-}
ightarrow \text{Na}_2\text{S}_1 = \text{Na}_2\text{S}$
Step11: Identify ions, cross for Li₂O
$\text{Li}^{1+} \text{O}^{2-}
ightarrow \text{Li}_2\text{O}_1 = \text{Li}_2\text{O}$
Step12: Identify ions, cross for CaO
$\text{Ca}^{2+} \text{O}^{2-}
ightarrow \text{Ca}_1\text{O}_1 = \text{CaO}$
Step13: Correct existing errors
- Magnesium Oxide: $\text{Mg}^{2+} \text{O}^{2-}
ightarrow \text{MgO}$ (charge written incorrectly as $\text{Mg}^{-2}$, fixed to $\text{Mg}^{2+}$)
- Cesium Fluoride: Anion is $\text{F}^{1-}$ (written incorrectly as $7^{-1}$, fixed), formula $\text{CsF}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| # | Name | Cation (+) | Anion (-) | Formula |
|---|---|---|---|---|
| 2 | Aluminum Chloride | $\text{Al}^{3+}$ | $\text{Cl}^{1-}$ | $\text{AlCl}_3$ |
| 3 | Aluminum Phosphide | $\text{Al}^{3+}$ | $\text{P}^{3-}$ | $\text{AlP}$ |
| 4 | Magnesium Oxide | $\text{Mg}^{2+}$ | $\text{O}^{2-}$ | $\text{MgO}$ |
| 5 | Cesium Fluoride | $\text{Cs}^{1+}$ | $\text{F}^{1-}$ | $\text{CsF}$ |
| 6 | Strontium Nitride | $\text{Sr}^{2+}$ | $\text{N}^{3-}$ | $\text{Sr}_3\text{N}_2$ |
| 7 | Lithium Sulfide | $\text{Li}^{1+}$ | $\text{S}^{2-}$ | $\text{Li}_2\text{S}$ |
| 8 | Calcium Chloride | $\text{Ca}^{2+}$ | $\text{Cl}^{1-}$ | $\text{CaCl}_2$ |
| 9 | Sodium Bromide | $\text{Na}^{1+}$ | $\text{Br}^{1-}$ | $\text{NaBr}$ |
| 10 | Beryllium Iodide | $\text{Be}^{2+}$ | $\text{I}^{1-}$ | $\text{BeI}_2$ |
| 11 | Strontium Fluoride | $\text{Sr}^{2+}$ | $\text{F}^{1-}$ | $\text{SrF}_2$ |
| 12 | Aluminum Fluoride | $\text{Al}^{3+}$ | $\text{F}^{1-}$ | $\text{AlF}_3$ |
| 13 | Potassium Nitride | $\text{K}^{1+}$ | $\text{N}^{3-}$ | $\text{K}_3\text{N}$ |
| 14 | Sodium Sulfide | $\text{Na}^{1+}$ | $\text{S}^{2-}$ | $\text{Na}_2\text{S}$ |
| 15 | Lithium Oxide | $\text{Li}^{1+}$ | $\text{O}^{2-}$ | $\text{Li}_2\text{O}$ |
| 16 | Calcium Oxide | $\text{Ca}^{2+}$ | $\text{O}^{2-}$ | $\text{CaO}$ |