QUESTION IMAGE
Question
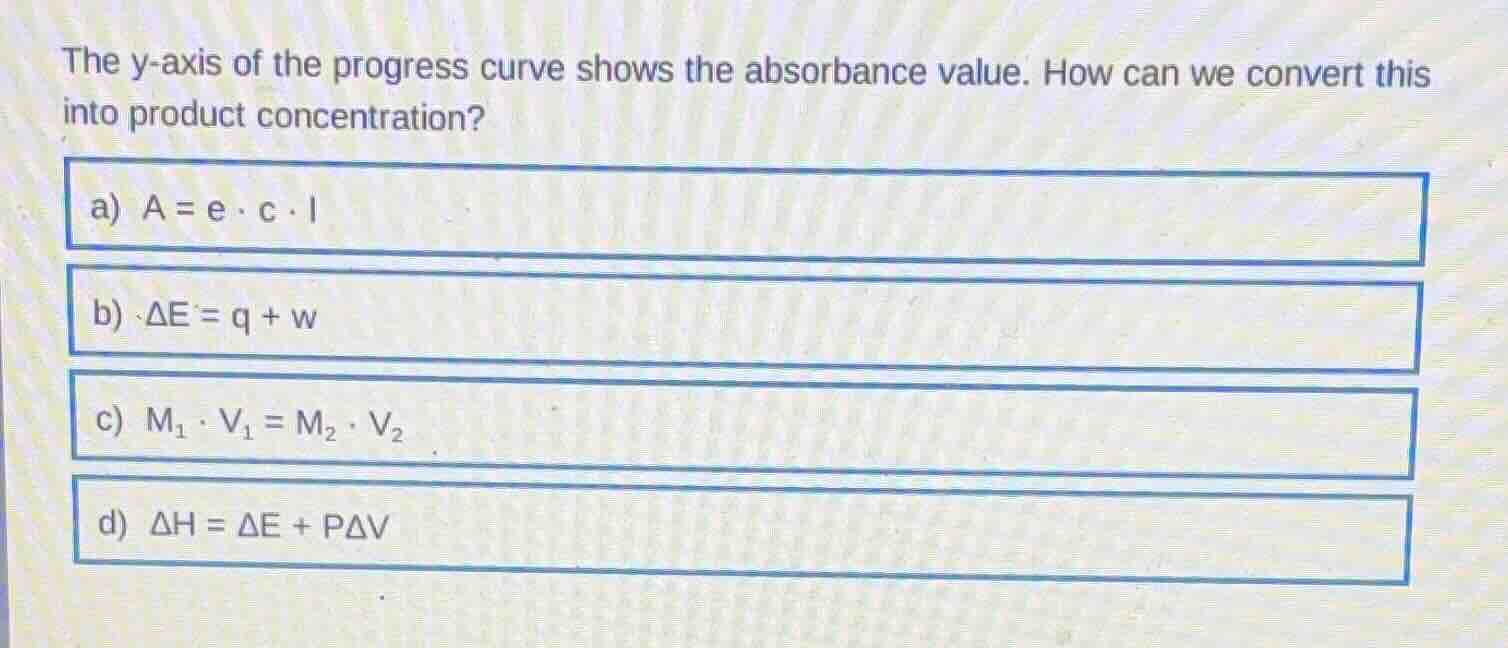
the y-axis of the progress curve shows the absorbance value. how can we convert this into product concentration?
a) $a = \varepsilon \cdot c \cdot l$
b) $\delta e = q + w$
c) $m_1 \cdot v_1 = m_2 \cdot v_2$
d) $\delta h = \delta e + p\delta v$
The question asks for the method to convert absorbance to product concentration. Beer-Lambert Law relates absorbance (A) to concentration (c), molar absorptivity (ε, written as e here), and path length (l). The other formulas relate to thermodynamics (b, d) and solution dilution (c), which are irrelevant to absorbance-concentration conversion.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
a) $A = e \cdot c \cdot l$