QUESTION IMAGE
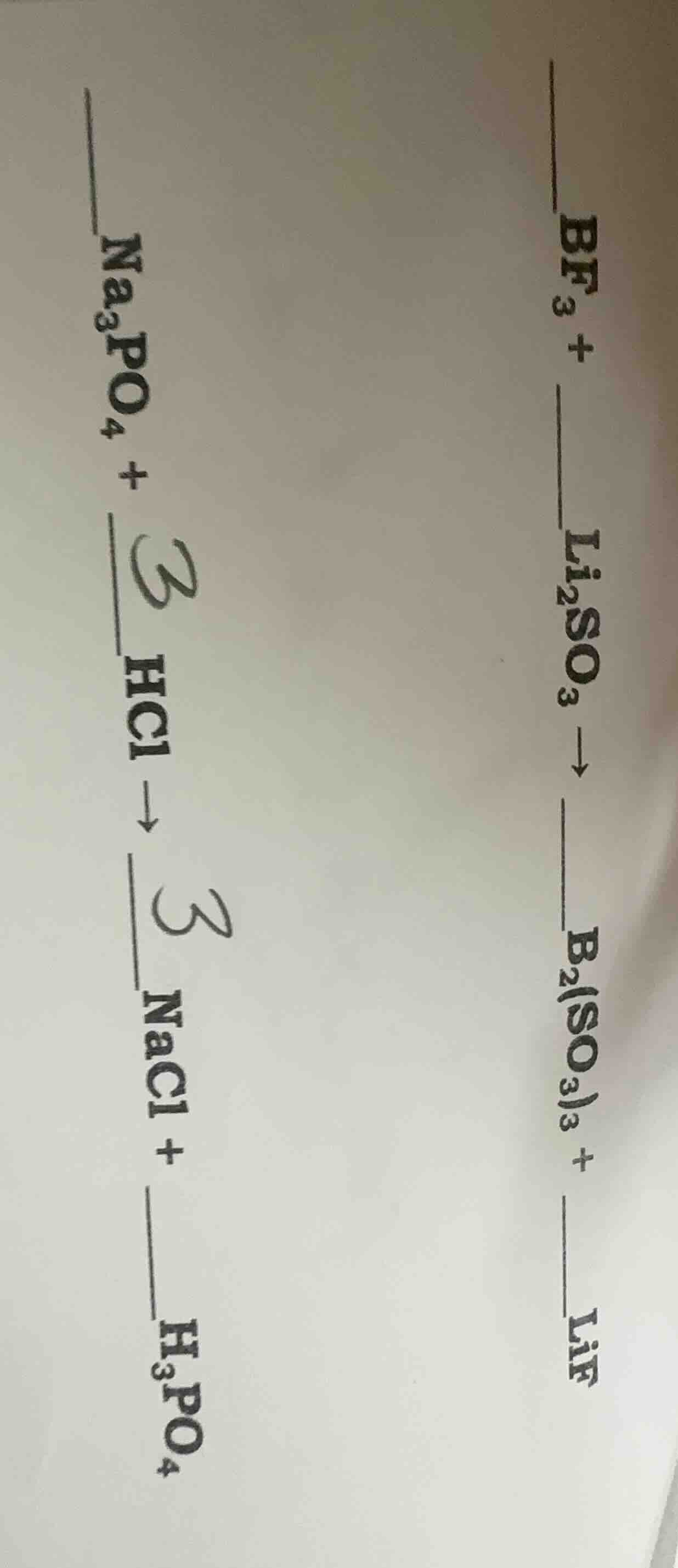
Question
bf₃ + __li₂so₃ → b₂(so₃)₃ + __lif
na₃po₄ + __hcl → nacl + __h₃po₄
Step1: Balance first reaction (B and F)
Start with balancing boron and fluorine.
$$2\text{BF}_3 + \text{Li}_2\text{SO}_3
ightarrow \text{B}_2(\text{SO}_3)_3 + 6\text{LiF}$$
Step2: Balance sulfur and lithium
Adjust coefficient for $\text{Li}_2\text{SO}_3$ to balance S and Li.
$$2\text{BF}_3 + 3\text{Li}_2\text{SO}_3
ightarrow \text{B}_2(\text{SO}_3)_3 + 6\text{LiF}$$
Step3: Verify first reaction
Check all atom counts: B=2, F=6, Li=6, S=3, O=9 on both sides.
Step4: Balance second reaction (Na and Cl)
Confirm Na and Cl are balanced, check remaining atoms.
$$\text{Na}_3\text{PO}_4 + 3\text{HCl}
ightarrow 3\text{NaCl} + \text{H}_3\text{PO}_4$$
Step5: Verify second reaction
Check all atom counts: Na=3, P=1, O=4, H=3, Cl=3 on both sides.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $2\text{BF}_3 + 3\text{Li}_2\text{SO}_3
ightarrow \text{B}_2(\text{SO}_3)_3 + 6\text{LiF}$
- $\text{Na}_3\text{PO}_4 + 3\text{HCl}
ightarrow 3\text{NaCl} + \text{H}_3\text{PO}_4$
Filled coefficients: 2, 3, 1, 6 (for first reaction); 1, 3, 3, 1 (for second reaction)