QUESTION IMAGE
Question
name _____________ period __ date ____
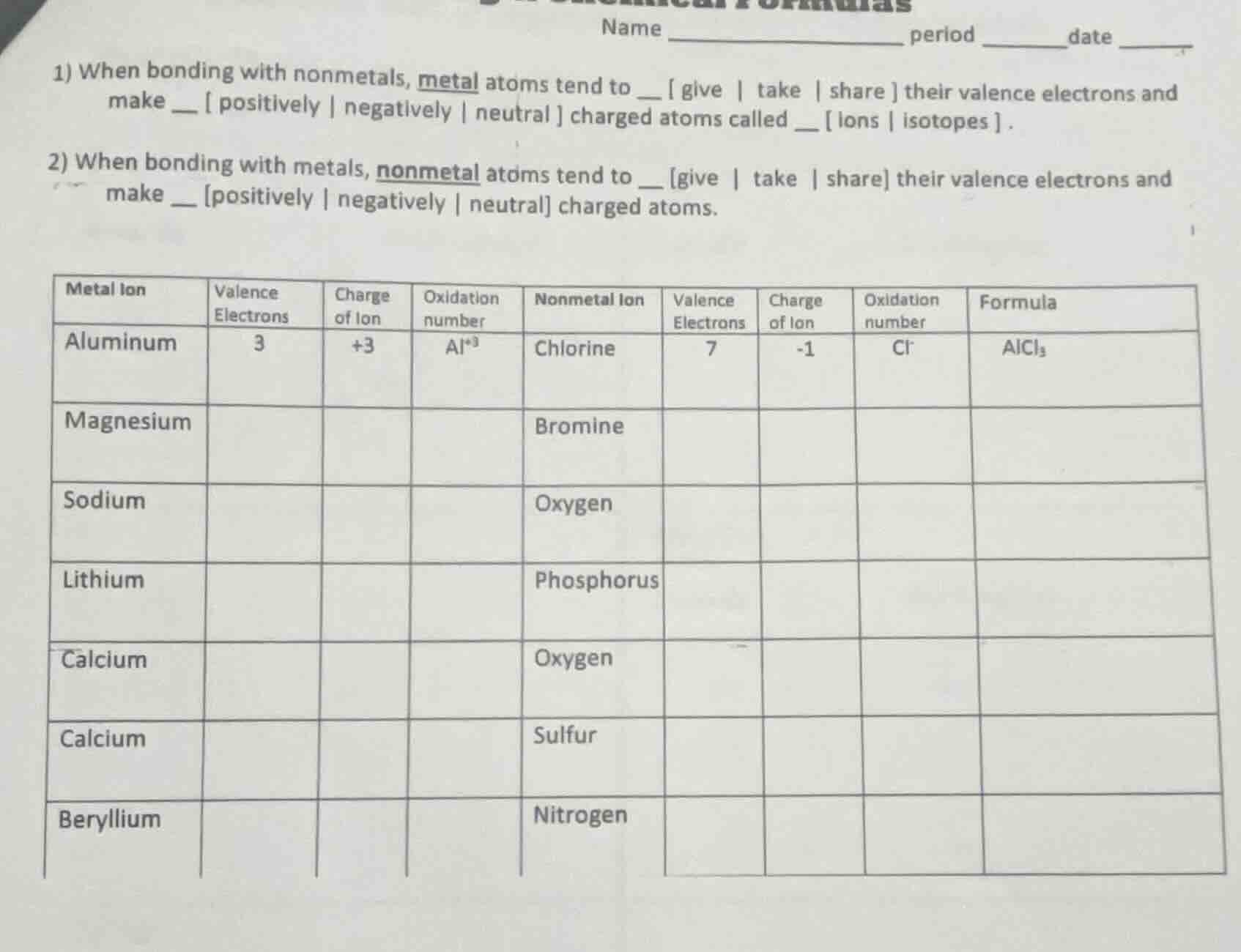
- when bonding with nonmetals, metal atoms tend to give | take | share their valence electrons and make positively | negatively | neutral charged atoms called __ ions | isotopes .
- when bonding with metals, nonmetal atoms tend to give | take | share their valence electrons and make positively | negatively | neutral charged atoms.
(chart: columns for metal ion, valence electrons, charge of ion, oxidation number, nonmetal ion, valence electrons, charge of ion, oxidation number, formula; rows with aluminum (3 valence e⁻, +3 charge, al⁺³, chlorine (7 valence e⁻, -1 charge, cl⁻, formula alcl₃), then magnesium, bromine; sodium, oxygen; lithium, phosphorus; calcium, oxygen; calcium, sulfur; beryllium, nitrogen)
Step1: Answer ionic bonding questions
Metals lose valence electrons to form positive ions (cations, a type of ion). Nonmetals gain these electrons to form negative ions (anions).
Step2: Fill metal ion properties
Use group numbers: Group 1 (Li, Na) has 1 valence electron, forms +1 ions; Group 2 (Mg, Ca, Be) has 2 valence electrons, forms +2 ions.
Step3: Fill nonmetal ion properties
Group 17 (Cl, Br) has 7 valence electrons, forms -1 ions; Group 16 (O, S) has 6 valence electrons, forms -2 ions; Group 15 (P, N) has 5 valence electrons, forms -3 ions.
Step4: Write neutral ionic formulas
Balance total positive/negative charge: e.g., Mg²⁺ + 2Br⁻ → MgBr₂; 3Li⁺ + P³⁻ → Li₃P.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- give; positively; ions
- take; negatively
| Metal Ion | Valence Electrons | Charge of Ion | Oxidation number | Nonmetal Ion | Valence Electrons | Charge of Ion | Oxidation number | Formula |
|---|---|---|---|---|---|---|---|---|
| Magnesium | 2 | +2 | Mg²⁺ | Bromine | 7 | -1 | Br⁻ | MgBr₂ |
| Sodium | 1 | +1 | Na⁺ | Oxygen | 6 | -2 | O²⁻ | Na₂O |
| Lithium | 1 | +1 | Li⁺ | Phosphorus | 5 | -3 | P³⁻ | Li₃P |
| Calcium | 2 | +2 | Ca²⁺ | Oxygen | 6 | -2 | O²⁻ | CaO |
| Calcium | 2 | +2 | Ca²⁺ | Sulfur | 6 | -2 | S²⁻ | CaS |
| Beryllium | 2 | +2 | Be²⁺ | Nitrogen | 5 | -3 | N³⁻ | Be₃N₂ |