QUESTION IMAGE
Question
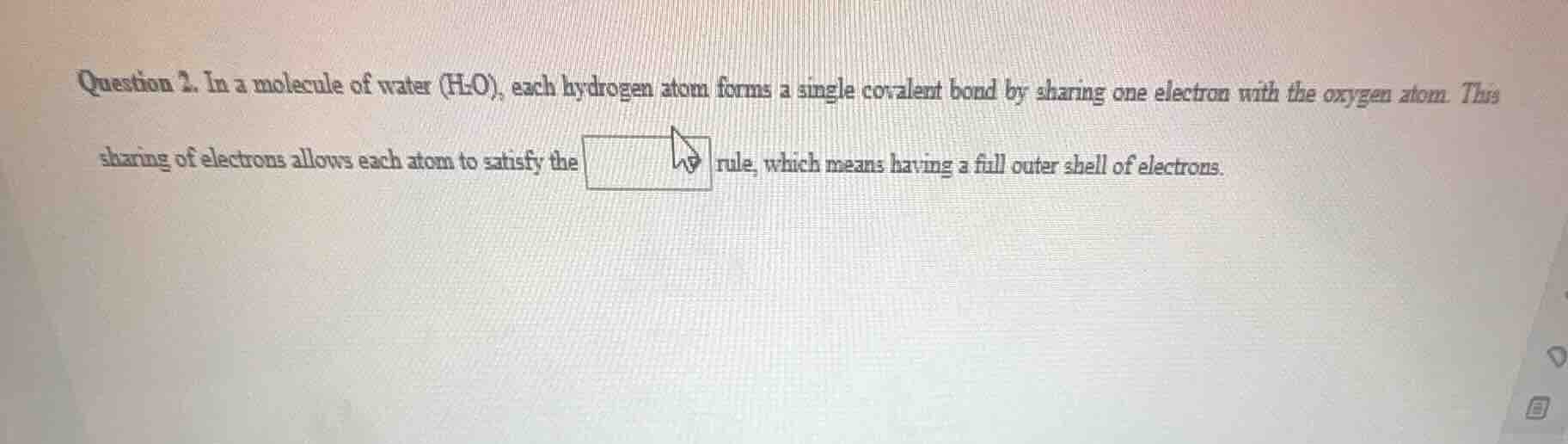
question 2. in a molecule of water (h₂o), each hydrogen atom forms a single covalent bond by sharing one electron with the oxygen atom. this sharing of electrons allows each atom to satisfy the rule, which means having a full outer shell of electrons.
In chemistry (a subfield of Natural Science), the octet rule (or duet rule for hydrogen) states that atoms tend to gain, lose, or share electrons to achieve a full outer electron shell (8 electrons for most, 2 for hydrogen like in H₂O). In H₂O, H shares one electron (achieving 2 in outer shell, duet) and O shares two (achieving 8, octet), so the rule here is the octet (or duet for H, but octet is the general term for this concept in bonding).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
octet (or duet, but octet is the standard rule referenced here for electron shell filling in covalent bonding, with hydrogen following a duet variation under the broader octet rule concept)