QUESTION IMAGE
Question
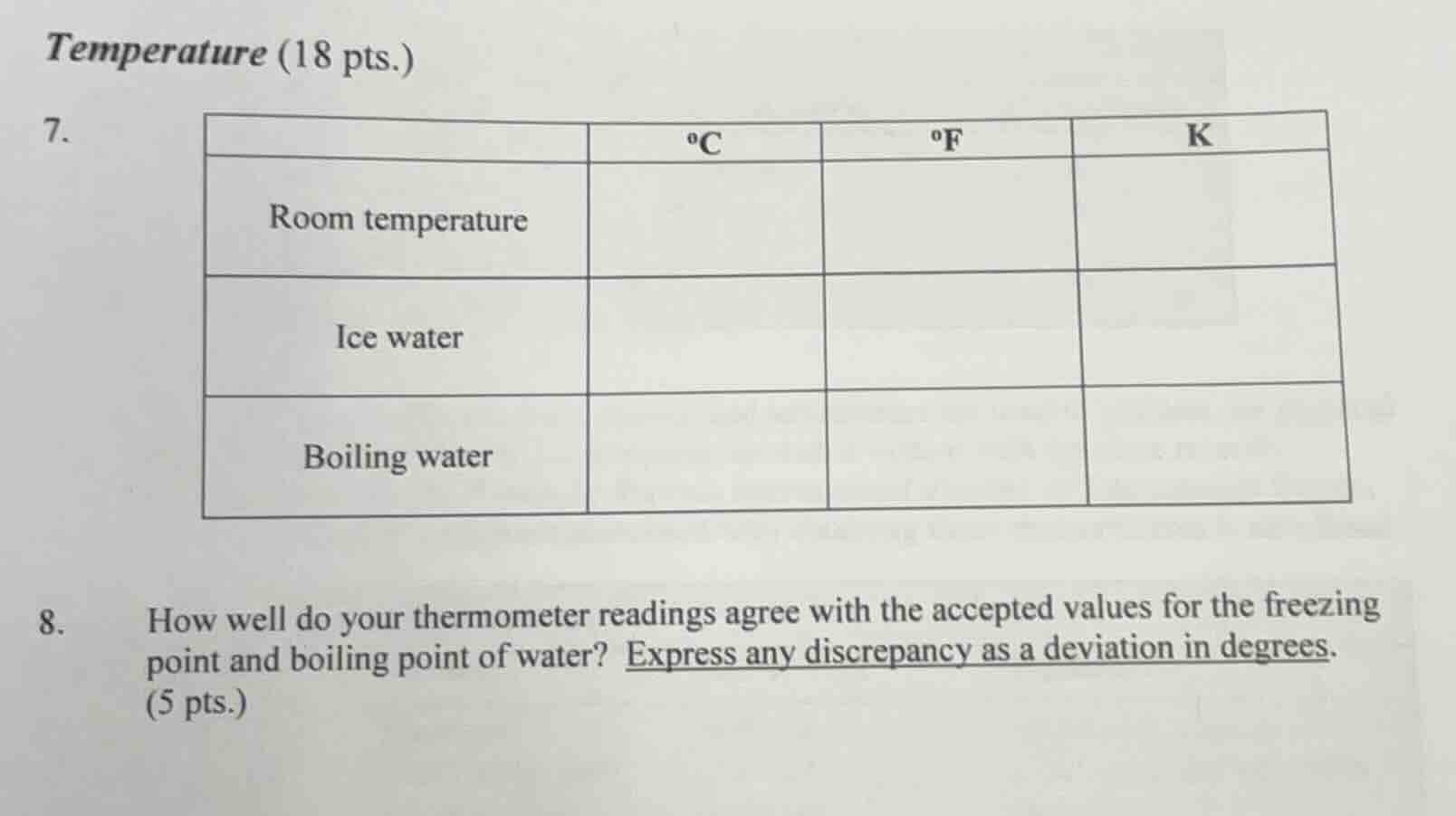
temperature (18 pts.)
7.
| °c | °f | k | |
| room temperature | |||
| ice water | |||
| boiling water |
- how well do your thermometer readings agree with the accepted values for the freezing point and boiling point of water? express any discrepancy as a deviation in degrees. (5 pts.)
Question 7: Filling the Temperature Table
Step 1: Recall Temperature Conversion Formulas
- Celsius to Fahrenheit: \( ^\circ\text{F} = \frac{9}{5}^\circ\text{C} + 32 \)
- Celsius to Kelvin: \( \text{K} = ^\circ\text{C} + 273.15 \)
Step 2: Room Temperature (Typical Values)
- \( ^\circ\text{C} \approx 25 \)
- \( ^\circ\text{F} = \frac{9}{5}(25) + 32 = 45 + 32 = 77 \)
- \( \text{K} = 25 + 273.15 = 298.15 \)
Step 3: Ice Water (Freezing Point of Water)
- \( ^\circ\text{C} = 0 \)
- \( ^\circ\text{F} = \frac{9}{5}(0) + 32 = 32 \)
- \( \text{K} = 0 + 273.15 = 273.15 \)
Step 4: Boiling Water (Boiling Point of Water)
- \( ^\circ\text{C} = 100 \)
- \( ^\circ\text{F} = \frac{9}{5}(100) + 32 = 180 + 32 = 212 \)
- \( \text{K} = 100 + 273.15 = 373.15 \)
Question 8: Analyzing Thermometer Readings
- Accepted Values:
- Freezing point of water: \( 0^\circ\text{C} \) (or \( 32^\circ\text{F} \), \( 273.15\text{K} \))
- Boiling point of water: \( 100^\circ\text{C} \) (or \( 212^\circ\text{F} \), \( 373.15\text{K} \))
- Deviation Calculation:
Deviation = \( |\text{Measured Value} - \text{Accepted Value}| \).
For example, if a thermometer reads \( 2^\circ\text{C} \) for ice water, the deviation is \( |2 - 0| = 2^\circ\text{C} \).
Final Answers
Question 7 Table:
| \( ^\circ\text{C} \) | \( ^\circ\text{F} \) | \( \text{K} \) | |
|---|---|---|---|
| Ice Water | 0 | 32 | 273.15 |
| Boiling Water | 100 | 212 | 373.15 |
Question 8:
To determine agreement, calculate the absolute difference between measured and accepted values (e.g., if ice water reads \( T_{\text{measured}} \), deviation = \( |T_{\text{measured}} - 0^\circ\text{C}| \) for Celsius, or similar for other scales). Report deviations in degrees (e.g., "My ice water reading deviated by \( 1^\circ\text{C} \) from \( 0^\circ\text{C} \), and boiling water deviated by \( 0.5^\circ\text{C} \) from \( 100^\circ\text{C} \)").
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Accepted Values:
- Freezing point of water: \( 0^\circ\text{C} \) (or \( 32^\circ\text{F} \), \( 273.15\text{K} \))
- Boiling point of water: \( 100^\circ\text{C} \) (or \( 212^\circ\text{F} \), \( 373.15\text{K} \))
- Deviation Calculation:
Deviation = \( |\text{Measured Value} - \text{Accepted Value}| \).
For example, if a thermometer reads \( 2^\circ\text{C} \) for ice water, the deviation is \( |2 - 0| = 2^\circ\text{C} \).
Final Answers
Question 7 Table:
| \( ^\circ\text{C} \) | \( ^\circ\text{F} \) | \( \text{K} \) | |
|---|---|---|---|
| Ice Water | 0 | 32 | 273.15 |
| Boiling Water | 100 | 212 | 373.15 |
Question 8:
To determine agreement, calculate the absolute difference between measured and accepted values (e.g., if ice water reads \( T_{\text{measured}} \), deviation = \( |T_{\text{measured}} - 0^\circ\text{C}| \) for Celsius, or similar for other scales). Report deviations in degrees (e.g., "My ice water reading deviated by \( 1^\circ\text{C} \) from \( 0^\circ\text{C} \), and boiling water deviated by \( 0.5^\circ\text{C} \) from \( 100^\circ\text{C} \)").