QUESTION IMAGE
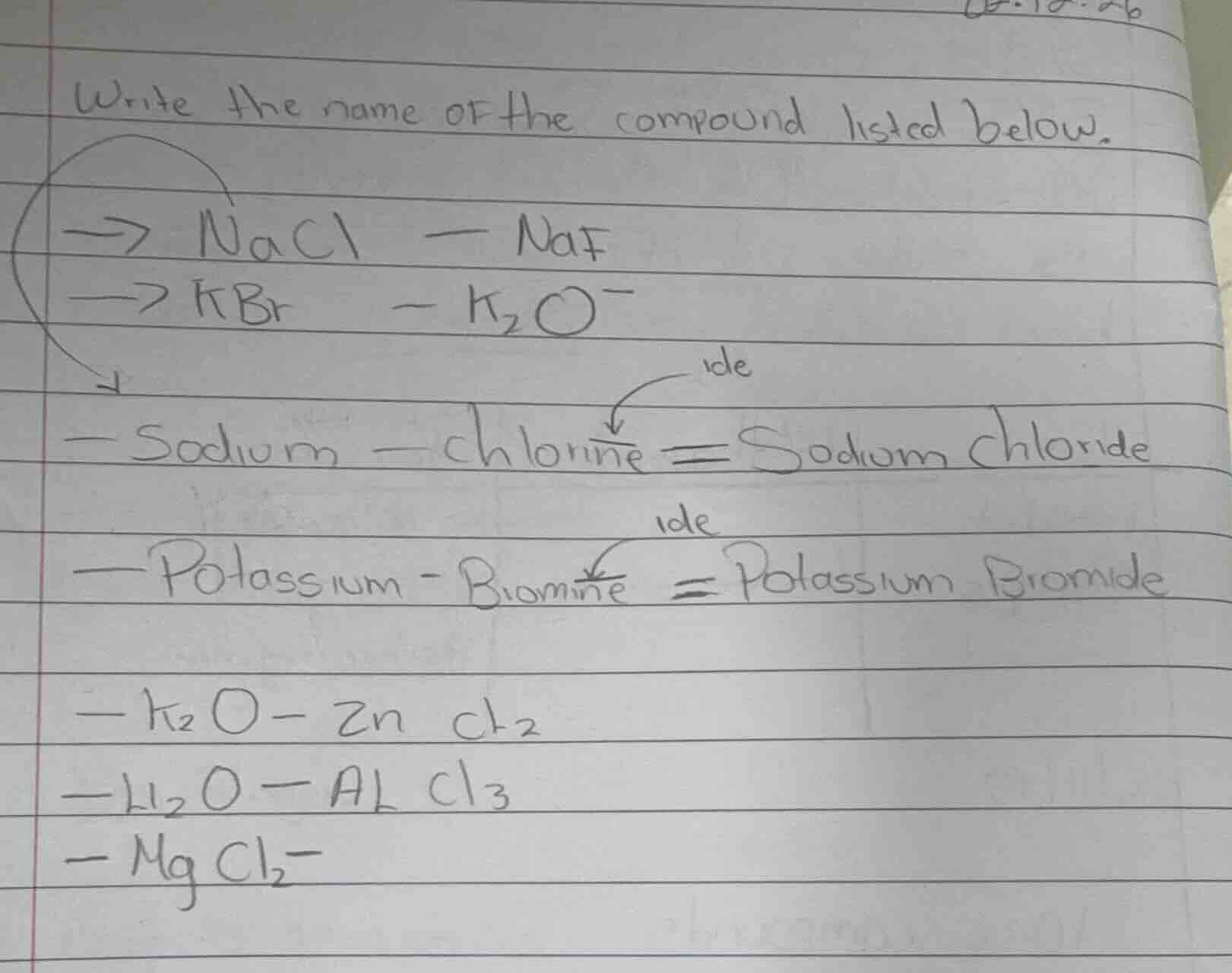
Question
write the name of the compound listed below.
-> nacl - naf
-> kbr - $k_2o$
- sodium - chlorine = sodium chloride
- potassium - bromine = potassium bromide
- $k_2o$ - $zncl_2$
- $h_2o$ - $alcl_3$
- $mgcl_2$
Brief Explanations
For ionic compounds, name the cation first (element name), then the anion with the suffix "-ide" replacing the end of the nonmetal/oxide name. For fixed-charge metals, no charge is needed in the name.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- NaF: Sodium Fluoride
- $K_2O$: Potassium Oxide
- $ZnCl_2$: Zinc Chloride
- $H_2O$: Water (or Dihydrogen Monoxide; common name is Water)
- $AlCl_3$: Aluminum Chloride
- $MgCl_2$: Magnesium Chloride